Coronary artery calcification and subclinical atherosclerosis: What's the score?
Coronary artery disease is the single most common cause of death in North America, but only 60% to 65% of acute myocardial infarctions or sudden cardiac deaths can be predicted using traditional Framingham risk assessment factors. Improved risk prediction may involve testing for coronary artery calcification, which is known to be an accurate marker for atherosclerotic disease and can be quantified with electron beam computed tomography and the newer multidetector CT techniques. Using this technology to screen symptomatic and asymptomatic patients can help to define risk and direct therapy, and may be a way to reduce the burden of atherosclerotic disease worldwide.
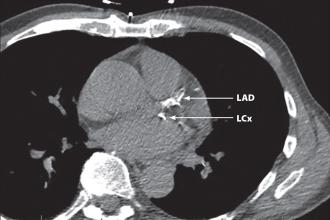
A CT scan assessment of the total burden of coronary atherosclerosis by CT scan can help clinicians manage symptomatic and asymptomatic patients.
Coronary artery disease (CAD) continues to be the number one cause of death in North America—a fact that is stimulating the search for screening techniques for its early detection. In 1999, 36% of all deaths in Canada were due to cardiovascular disease, with 54% of cardiovascular deaths, or over 42 000 cases, due to CAD.[1] CAD is the major cause of death in women in North America, and in 1997 10 times more women died from CAD than from breast cancer.[2] Health Canada reported in 1998 that the economic burden of cardiovascular disease to the Canadian economy was approximately $18.5 billion, more than for any other disease.[1]
Primary prevention of CAD requires identification of an at-risk population and, more important, at-risk individuals, so that effective intervention can be implemented. Unfortunately, traditional Framingham risk assessment factors predict only 60% to 65% of hard cardiac events—acute myocardial infarction (MI) or sudden cardiac death.[3] In up to 50% of patients with CAD the first manifestation of the disease is MI or death,[3] exemplifying the acute need to improve risk prediction.
The preliminary results of the Inter-Heart Study[4] suggest that when additional risk factors such as stress, sedentary lifestyle, and unhealthy diet are added to traditional Framingham risk assessment factors, up to 90% of MIs could be predicted. These additional risks can, however, be very difficult to measure accurately and their utility in the clinical environment is not yet proven. Because we know that atherosclerosis infiltrates the arterial wall long before it causes vessel obstruction and produces symptoms,[5] earlier identification of this process should be part of risk prediction. And since atherosclerosis is a diffuse disease rather than a focal one, assessment for burden of subclinical coronary artery disease should evaluate the vessel wall throughout the coronary arterial tree.
Role of calcium in athersclerotic plaque
Coronary artery calcification (CAC) is a linear estimate of the total burden of coronary atherosclerosis that highly correlates with autopsy and intravascular ultrasound assessment.[3] In fact, CAC provides a better estimate of burden of disease than luminal stenosis as determined by angiography,[6] since less-obstructive plaques actually give rise to more occlusions than more-obstructive plaques because of their greater number. In fact, 70% of patients with acute MI have stenoses of less than 50% in their culprit artery on angiography.[7] Therefore, stress testing (treadmill, nuclear medicine, or echo), which typically diagnoses the physiological effects of high-grade stenosis, will fail to identify a significant number of persons at risk for a hard event.[8] CAC detection by CT directly detects only hard or calcified plaque, but the calcification found essentially serves as a surrogate marker for soft or noncalcified plaque as well, since in the vast majority of patients both types of plaques coexist proportionally.[9] It should be noted, however, that no current imaging test is able to identify plaque that is prone to rupture, and that the presence of calcification in a coronary artery does not necessarily indicate a stenosis at that site; the value of CAC assessment is, rather, in its ability to reflect the overall burden of coronary athersclerosis.
It has long been known that CAC observed during fluoroscopy at the time of cardiac catheterization in symptomatic patients carries prognostic significance for hard events independent of age, gender, and angiographic identification of diseased vessels.[10] Because fluoroscopy can be compromised by patient habitus and overlying anatomic structures, it is relatively insensitive in detection of CAC, and in the 1980s, electron beam computed tomography (EBCT) replaced this technique. The major advantages of EBCT are its fast acquisition time (approximately 100 ms/slice) and low-dose radiation (0.7 mSv). In addition, an extensive literature has developed over the last two decades regarding this technique. The disadvantages of EBCT include its high cost and limited ability to adjust parameters to optimize image quality.
Over the last 10 years, major advances have been made in CT scan technology, allowing the advent of multislice CT, in which multiple imaging slices can be acquired simultaneously, proportional to the number of detectors (ranging from 4 to 64 detectors in currently available systems). Using prospective ECG-gated techniques and optimized reconstruction algorithms, effective scan times of 200 to 300 ms can be obtained. The advantages of these systems include their lower cost compared with EBCT, better spatial resolution, and potential for parameter adjustment; disadvantages include a slightly higher radiation dose (1.3 mSv) and possible motion artifacts in patients with high resting heart rates.
The extensive literature from EBCT studies has generated considerable interest in comparing multislice CT with EBCT for CAC scoring in order to use the existing EBCT database and not repeat a large number of scientific studies. Although there remains some controversy, most studies of patients with heart rates lower than 80 bpm have concluded that there is high correlation between CAC scoring using these two techniques, with r values ranging from 0.96[11] to 0.99[12]. From the point of view of reproducibility of CAC scoring, both electron beam and multislice methods have low intrascan and intraobserver variability. Interscan accuracy is obviously desirable for follow-up, including for those patients receiving treatment, but remains problematic with both methods, and measures approximately 10% to 14%.[13]
No special preparation for CAC scoring is required, other than abstinence from caffeine on the day of the scan in order to ensure an optimized resting heart rate of 80 bpm. ECG electrodes are applied, scan preparation is completed, and the acquisition scan is performed, requiring only a 20-s breath-hold. The 45 to 65 images obtained are analyzed by a radiologist on a workstation equipped with specialized software that yields a total calcium score (Agatston-Janowitz score), which can range from zero to several thousand. This total score is based on the scores of the five interrogated coronary arteries (left main, left anterior descending, left circumflex, right coronary, and posterior descending), which in turn represent the total number of calcified plaques in each artery. Each plaque is scored based on the lesion area and density.[14] The total calcium score is then adjusted for age and gender using tables from a large database. Calcium volume scores are also generated, which may be more useful for patient follow-up in the future.[13] The other anatomic regions included in the scan must also be carefully assessed, as in one recent study[15] 8% of patients had an extra-cardiac abnormality that required follow-up. In some centres, subjects are afforded an immediate after-scan interview with a physician, who reviews not only the results of the scan but also the images from it. This protocol has been shown to improve patient compliance in modifying pre-existing cardiac risk factors.[16]
CAC scoring in symptomatic patients
A CAC score of zero in a technically acceptable examination makes the presence of atherosclerotic plaque, including unstable plaque, very unlikely, with a negative predictive value of approximately 95%, resulting in a low risk of a cardiovascular event in the next 2 to 5 years.[17] This high negative predictive value can be useful in a number of clinical scenarios. Studies in the emergency room setting have shown a zero score to be highly accurate in excluding coronary causes for chest pain.[18] A zero score may also be helpful in excluding coronary causes in low-risk patients presenting with atypical chest pain. Thus, the exclusion of significant CAC and atherosclerosis may be used to determine the need for hospital admission or invasive diagnostic procedures.
In patients with ischemic cardiomyopathy, clinical manifestations are often indistinguishable from those seen in primary dilated cardiomyopathy, and in this specific cohort the presence of CAC has been associated with a 99% sensitivity for ischemic cardiomyopathy.[3]
Stress testing is a relatively inexpensive method to risk-stratify in symtomatic patients. However, in patients with a low risk treadmill test, the test itself is an imprecise measurement of atherosclerotic burden, with sensitivity of 68% and a widely variable false-positive rate, ranging from 0% to 83%,[19] that can lead to more expensive and invasive diagnostic testing. In one study, the absence of CAC as confirmed by EBCT reliably identified symptomatic patients with a false-positive treadmill test,[19] with a negative predictive value for a zero score of 93%, compared with an angiographic gold standard. In symptomatic patients it may, therefore, make sense to consider combining a noninvasive morphological test such as CAC testing with a noninvasive physiological test such as exercise treadmill testing before embarking on invasive diagnostic procedures.
Patients who do not benefit from CAC scoring include patients with chest pain typical of CAD, patients at high clinical risk, such as insulin-dependent diabetics, and patients with a known diagnosis of CAD.
CAC scoring in asymptomatic patients
The role of CAC scoring in risk prediction for asymptomatic patients has recently been the subject of increasingly large studies. In a large observational study following more than 10 000 asymptomatic patients, Shaw and colleagues demonstrated conclusively that CAC scoring added prognostic information over and above that derived from traditional Framingham risk assessments.[20] In the St. Francis Heart Study,[21] which followed more than 5000 subjects for more than 4 years, a CAC score of over 100 predicted all atherosclerotic cardiovascular events and was associated with a tenfold increase in risk of such events in the presence of CAC. In an older study, Raggi and colleagues[22] followed 632 asymptomatic subjects with risk factors for CAD and reported a zero CAC score to be associated with a 0.11% per year hard event rate and a CAC score over 400 to be associated with a 4.8% per year event rate. The event rate in patients with CAC scores in the highest quartile was 22 times the event rate in patients with CAC scores in the lowest quartile. Furthermore, CAC scoring provided incremental prognostic value when added to conventional risk assessment for CAD. Greenland and colleagues[23] further demonstrated that CAC scoring can be used to significantly modify predicted risk obtained from the Framingham risk score alone, and could thereby alter clinical decision making. Kondos[24] followed 5635 initially asymptomatic low-risk to intermediate-risk patients for more than 3 years and demonstrated that the presence of CAC was the best predictor of hard events (relative risk [RR] = 5.8) when compared with traditional risk factors such as smoking (RR = 1.2) and diabetes (RR = 1.2) in men. Of interest is that all these studies were published some time after the American Heart Association/American College of Cardiology consensus document of July 2000,[17] which cautions against the use of coronary artery calcification scoring for diagnosing obstructive coronary artery disease because of low specificity.
Two additional studies of note followed specific cohorts of patients. Wayhs and colleagues[25] followed patients with very high calcium scores of over 1000 for an average of 17 months and found that 36% of patients suffered a hard cardiac event, corresponding to an annualized event rate of 25%, underlining the dramatic prognostic significance of such scores. The second study[26] followed 102 patients (mean age 41) with EBCT performed within 1 to 14 days of the first unheralded acute myocardial infarction, to address the question of whether coronary events in younger individuals might not be predicted by CAC scoring because of the role played by rupture of more lipid-rich, less heavily calcified plaques. When compared with controls who were matched for sex, age, and traditional risk factors, more than 60% of patients with hard events scored above the 90th percentile for CAC scoring, compared with only 5.8% of controls.
Management of patients with positive CAC scans
Coronary calcification is an unequivocal marker for atherosclerotic disease, with multiple studies demonstrating the sensitivity of CAC scoring to be in the range of 92% to 100% for occlusive CAD.[19] Bielak and colleagues[27] demonstrated the specificity for CAC scoring in predicting greater than 50% coronary artery stenosis using an angiographic gold standard to be 72%, when corrected for verification bias. Unfortunately, most studies have measured specificity based on this angiographic gold standard. As discussed previously, this approach introduces bias against CAC scoring, as the clinically more important less-obstructive plaques demonstrating calcium in the arterial wall are not as accurately assessed with angiography as are the less prevalent more-obstructive plaques. Nevertheless, based on published data, so-called false-positives from CAC scoring will occur compared with the angiographic gold standard, and subsequent workup schemes need to accommodate this effect.
A positive CAC scan indicates the presence of atherosclerosis and confirms the diagnosis of coronary artery disease. There is, however, a need to distinguish between coronary artery disease and ischemic heart disease, and the next step is to determine the presence or absence of ischemia with an exercise treadmill test or myocardial perfusion scan. Wong and colleagues[28] demonstrated a statistically significant increased risk of cardiac events in those patients whose CAC scores were above the 50th percentile, adjusted for age and gender. This cut-off point of the 50th percentile has been confirmed by other investigators.[29,30] We therefore recommend that patients scoring over this adjusted median calcium score undergo physiological testing, such as exercise treadmill testing or myocardial perfusion scans. Elevated C-reactive protein levels also indicate a twofold risk of coronary events after adjustment for demographic and prior risk factors.[31] The determination of C-reactive protein in patients with CAC scoring above the 50th percentile provides another method of risk stratification, as CAC scoring and C-reactive protein have been shown to be complementary for prediction of cardiovascular events.[31]
If definite myocardial ischemia is demonstrated either clinically or with functional testing, such as exercise treadmill testing or myocardial perfusion scans, there is very good evidence that the patient should be treated with a statin, irrespective of baseline total cholesterol or LDL levels.[32]
Although it makes sense to lower cholesterol levels in a patient with CAD in the absence of ischemia, there has been no prospective randomized controlled trial published as yet to corroborate this widely held belief. It is, however, under intense study, and prevailing opinion holds that the direct visualization of atherosclerosis in the coronary tree is a better predictor of an event than the inference of disease based purely on a risk profile. In a patient at moderate risk for a future event, the presence of atherosclerosis in the coronary arteries should, in our opinion, trigger an intensification of risk modification, including more aggressive lipid control. In contrast, a CAC score of zero may allow downstaging of risk stratification and less aggressive risk modification.
One of the most important aspects of CAC scoring is optimized communication between the CT scan clinic, the patient, and the physician (or physicians) involved. Direct physician consultation after the CAC scoring and a review of images with patients can reassure those with positive results but low scores, and encourage compliance in those requiring lifestyle modification and pharmacological intervention.[16,33] It is also important that the CT scan clinic develop a close relationship with cardiologists, who can further advise the family physicians and patients regarding appropriate workup of patients with positive CAC scans. One point remains clear: in an asymptomatic patient a high CAC score only very rarely should result in an expedited coronary angiogram without further noninvasive physiological testing. It is also clear that both Framingham risk assessment and CAC scoring should be undertaken simultaneously to predict risk, as their effects are additive.[34] Further elucidation of the role of CAC scoring and, indeed, other noninvasive tests such as carotid intima-media thickness testing, will be forthcoming at the completion of the Multi-Ethnic Study of Atherosclerosis (MESA) sponsored by the NIH.
The radiation dose for a typical CAC scoring examination is 1.3 mSv, which must be compared with background radiation exposure in Canada that is approximately 2 to 3 mSv per year.[33] Although a detailed review of the effects of low-level radiation is beyond the scope of this article, there is general agreement that the risk of radiation- induced cancer from any source, however small, decreases with advancing age. For this reason, CT screening for CAD should be limited to patients older than 40 (exceptions include those with a strong family history of premature CAD). Similarly, since risk predictions from CAC scoring are based on a 2- to 5-year interval, it is not appropriate to routinely scan patients on an annual basis. Although some early studies have examined longitudinal assessment of CAC scores in patients receiving statin therapy,[35] sample sizes are too small in these studies to currently recommend CAC scoring for assessment of coronary atherosclerosis in patients on lipid-lowering therapies. Until such studies are completed, there is no proven benefit to CAC scoring in patients with known CAD.
CAC scoring is best applied as a screening test in a population with intermediate pretest probability of disease,[36] which in the US amounts to approximately 36 million people who are considered at intermediate risk according to the National Cholesterol Education Program (NCEP) Adult Treatment Panel III.[5] As of 2001, the NCEP now specifically recommends the use of CAC scoring to assist in risk stratification for these intermediate-risk patients and the elderly, in whom traditional risk factors lose some of their predictive power.[37] European guidelines also support the combined use of CAC scoring and Framingham risk assessment.[38] The Society of Athersclerosis Imaging has recommended CAC scoring in primary prevention for men 35 years of age and older, and for women 45 years of age and older if any traditional risk factors are present. In the absence of risk factors, scanning is still recommended for those 10 years older than the recommended age.[39] A new American Heart Association statement on CAC is pending.[40]
Current attitudes and policies regarding cardiovascular screening do not support routine atherosclerotic imaging[5] and further large-scale studies, such as MESA, and the long-term outcome data they can provide, will be important. However, a preliminary report of more than 5000 asymptomatic patients stratified into clinical low-risk, intermediate-risk, and high-risk groups found 13% of the low-risk group to have significant CAC.[41] This result, combined with well-established data indicating that traditional Framingham risk assessment predicts only 60% to 65% of hard cardiac events,[3] raises the question of using CAC scoring to screen subjects who appear to be at low risk based on these traditional risk factors. Nevertheless, CAC scoring of patients in this group remains controversial, as does self-referral.[33]
Among the disadvantages of screening for subclinical atherosclerosis are the limited availability of atherosclerosis imaging and the high cost of noninvasive testing. Although some modeling has been performed suggesting that CAC scoring could be cost-effective in both symptomatic[42] and asymptomatic[5] patients, much more analysis is needed using data from prospective randomized controlled trials.
Atherosclerosis and coronary artery disease are huge health concerns in Canada and North America. They constitute the largest single cause of death and exact a financial burden of billions of dollars annually. It is well known that lifestyle modification and drug therapy in selected individuals can reduce the risk of hard cardiac events, but current Framingham risk assessment is suboptimal. Coronary artery calcification as measured by CT is the most powerful independent predictor of hard cardiac events, and the introduction of multidetector CT allows cheaper and more accessible scans than EBCT. According to current guidelines from some agencies, CAC scoring should be considered to increase the accuracy of risk stratification for intermediate-risk individuals and to direct further testing and treatment. As primary prevention represents the only hope of reducing the burden of atherosclerosis worldwide, CAC scoring and other imaging and biochemical risk predictors will likely play an increasingly important role in health care management in the coming years.
Competing interests
Dr Forster receives a salary from Canada Diagnostic Centres (BC), where he is medical director. Coronary artery calcification studies are performed at this private clinic. Neither he nor any member of his practice group has an equity position with Canada Diagnostic Centres.
Acknowledgments
The authors are grateful for the assistance of Ms Marilyn Stuart in typing this manuscript.
References
1. Heart and Stroke Foundation of Canada. www.heartandstroke.ca (accessed 9 August 2004).
2. American Heart Association 1999 Heart and Stroke Statistical Update. Dallas, Texas: American Heart Association; 1998.
3. Budoff MJ. Atherosclerosis imaging in calcified plaque: Coronary artery risk assessment. Prog Cardiovasc Dis 2003;46:135-148. PubMed Abstract Full Text
4. Yusef S. Inter-heart: A study of risk factors for first myocardial infarction in 52 countries and over 27,000 subjects [Program #104]. Presented at European Society of Cardiology Congress; 29 August 2004; Munich, Germany. www.escardio.org/knowledge/congress/clinical (accessed 14 March 2005).
5. Shaw LJ, Raggi E, Burman DS, et al. Cost effectiveness for screening of cardiovascular disease with measures of coronary calcium. Prog Cardiovasc Dis 2003;46:171-184. PubMed Abstract Full Text
6. Sangiorgi G, Rumberger JA, Severson A, et al. Arterial calcification and not lumen stenosis is highly correlated with atherosclerotic plaque burden in humans: A histologic study of 723 coronary artery segments using non-delcacifying methodology. J Am Coll Cardiol 1998;31:126-133. PubMed Abstract Full Text
7. Falk E, Shah EK, Fuster V. Coronary plaque disruption. Circulation 1995;92:657-671. PubMed Citation Full Text
8. He ZX, Hedrick TD, Pratt CM, et al. Severity of coronary artery calcification by electron beam computed tomography predicts silent myocardial ischemia. Circulation 2000;101:244-251. PubMed Abstract Full Text
9. Schmermund A, Baumgart D, Adamzik M, et al. Comparison of electron beam computed tomography and intracoronary ultrasound detecting calcified and non-calcified plaque in patients with acute coronary syndromes and no or minimal to moderate angiographic coronary disease. Am J Cardiol 1998;81:141-146. PubMed Abstract
10. Margolis JR, Chen JT, Kong Y, et al. The diagnostic and prognostic significance of coronary artery calcification: A report of 800 cases. Radiology 1980;137:609-616. PubMed Abstract
11. Stanford W, Thompson BH, Burns TL, et al. Coronary artery calcium quantification at multi-detector row helical CT vs electron beam CT. Radiology 2004;30:397-402. PubMed Abstract Full Text
12. Knez A, Becker C, Becker A, et al. Determination of coronary calcium with multi-slice spiral computed tomography: A comparative study with electron-beam CT. Int J Card Imaging 2002;18:295-303. PubMed Abstract
13. Hong C, Bae KT, Pilgram TK. Coronary artery calcium: Accuracy and reproducibility of measurements with multi-detector row CT—Assessments of effects of different thresholds in quantification methods. Radiology 2003;227:795-801. PubMed Abstract Full Text
14. Agatston AS, Janowitz WR, Hildner FJ, et al. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 1990;15:827-832. PubMed Abstract
15. Horton KM, Post WS, Blumenthal RS. Prevalence of significance of non-cardiac findings on electron beam computed tomography coronary artery calcium screen examinations. Circulation 2002;106:532-534. PubMed Abstract Full Text
16. Wong ND, Detrano RC, Diamond G, et al. Does coronary artery screening by electron beam computed tomography motivate potentially beneficial lifestyle behaviors? Am J Cardiol 1996;78:1220-1223. PubMed Abstract
17. O’Rourke RA, Brundage BH, Froelicher VF, et al. American College of Cardiology/American Heart Association expert consensus document on electron beam computed tomography for the diagnosis and prognosis of coronary artery disease. Circulation 2000;102:126-140. PubMed Citation Full Text
18. Laudon DA, Vukov LF, Breen JF, et al. Use of electron beam computed tomography in the evaluation of chest pain in the emergency department. Ann Emerg Med 1999;33:15-21. PubMed Abstract Full Text
19. Lamont DH, Budoff MJ, Shavelle DM, et al. Coronary calcium scanning adds incremental value to patients with positive stress tests. Am Heart J 2002;143:861-867. PubMed Abstract Full Text
20. Shaw LJ, Raggi P, Schisterman E, et al. Prognostic value of cardiac risk factors and coronary artery calcium screening for all-cause mortality. Radiology 2003;228:826-833. PubMed Abstract Full Text
21. Arad Y, Roth M, Newstein D, et al. Coronary calcification, coronary risk factors, and atherosclerotic cardiovascular disease events. The St. Francis Heart Study. J Am Coll Cardiol 2003;41:6-7.
22. Raggi P, Cooil B, Callister TQ. Use of electron beam tomography to develop models for prediction of hard coronary events. Am Heart J 2001;141:375-382. PubMed Abstract
23. Greenland P, LaBree L, Azen SP, et al. Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals. JAMA 2004;291:210-215. PubMed Abstract Full Text
24. Kondos GT, Hoff JA, Sevrukov A, et al. Electron-beam tomography coronary artery calcium and cardiac events: A 37-month follow-up of 5635 initially asymptomic low- to intermediate-risk adults. Circulation 2003;107:2571-2576. PubMed Abstract Full Text
25. Wayhs R, Zelinger A, Raggi P. High coronary artery calcium scores pose an extremely elevated risk for hard events. J Am Coll Cardiol 2002;39:225-230. PubMed Abstract
26. Pohle K, Ropers D, Maffert R, et al. Coronary calcification in young patients with first unheralded myocardial infarction: A risk factor matched analysis by electron beam tomography. Heart 2003;89:625-628. PubMed Abstract Full Text
27. Bielak LF, Rumberger JA, Sheedy PF, et al. Probabilistic model for prediction for angiographically defined obstructive coronary artery disease using electron beam computed tomography calcium score strata. Circulation 2000;102:380-385. PubMed Abstract Full Text
28. Wong ND, Budoff MJ, Pio J, et al. Coronary calcium and cardiovascular event risk: Evaluation by age and sex specific quartiles. Am Heart J 2002;143:456-459. PubMed Abstract Full Text
29. O’Malley PJ, Taylor A, Jackson JL, et al. The prognostic value of coronary electron beam computed tomography for myocardial infarction and cardiac death: A systematic review and metaanalyis. Am J Cardiol 2000;85:945-948.
30. Wong ND, Shu JC, Detrano RC, et al. Coronary artery calcium evaluation by electron beam computed tomography and its relationship to new cardiovascular events. Am J Cardiol 2000;86:495-498. PubMed Abstract
31. Park R, Detrano R, Xiang M. Combined use of computed tomography coronary calcium scores and C-reactive protein levels in predicting cardiovascular events in nondiabetic individuals. Circulation 2002;106:2073-2077. PubMed Abstract Full Text
32. Heart Protection Study Collaborative Group: MRC/BHF heart protection study of cholesterol lowering with sinvastatin in 20,536 high risk individuals: A randomized placebo controlled trial. Lancet 2002;360:7-22. PubMed Abstract
33. Forster BB, Mayo JR. Rational computed tomography screening in 2003. Can Assoc Radiol J 2003;54:14-17. PubMed Citation
34. Thompson GR, Partridge J. Coronary calcium score: The coronary risk impact factor. Lancet 2004;363:557-559. PubMed Abstract
35. Achenbach S, Ropers D, Pohle K, et al. Influence of lipid-lowering therapy on progression of coronary artery calcification: A prospective evaluation. Circulation 2002;106:1077-1082. PubMed Abstract Full Tex
36. Raggi P. Role of coronary calcium screening in preventive cardiology. Prev Cardiol 2003;6:214-217. PubMed Abstract Full Text
37. National Institutes of Health. Third report of the national cholesterol education program expert panel on detection, evaluation and treatment of high blood cholesterol in adults. (Adult treatment panel III). Final report. NIH Publication No.02-5215, September 2002. www.nhlbi.nih.gov/guidelines/cholesterol/atp3_rpt.htm (accessed 5 February 2005).
38. De Backer G, Ambrosionie E, Borch-Johnsen K, et al. Executive summary: European guidelines on cardiovascular disease prevention in clinical practice. Third joint task force of European and other societies on cardiovascular disease prevention in clinical practice. Eur Heart J 2003;24:1601-1610. PubMed Citation Full Text
39. Hecht HS. Translating tomographic plaque imaging into treatment: Interventional lipidology. Prog Cardiovasc Dis 2003;46:149-170. PubMed Abstract Full Text
40. Hecht HS. Introduction: Tomographic plaque imaging 2003. Prog Cardiovasc Dis 2003;46:113.
41. Desai M, Nasir K, Brounstein JB, et al. Framingham risk estimate is weakly correlated with coronary artery calcification in asymptomatic population [#845-2]. Presented at the American College of Cardiology meeting; March 2004; New Orleans, USA. www.acc04online.acc.org (accessed 14 March 2005).
42. Rumberger JA, Behrenbeck T, Breen JF, et al. Coronary calcification by electron beam computed tomography and obstructive coronary artery disease: A model for costs and effectiveness of diagnosis as compared with conventional cardiac testing methods. J Am Coll Cardiol 1999;33:453-462. PubMed Abstract
Bruce B. Forster, MD, FRCPC, Saul Isserow, MD, FRCPC
Dr Forster is an associate professor in the Department of Radiology, Faculty of Medicine, at UBC. He is also a staff radiologist at UBC Hospital and medical director of Canada Diagnostic Centres (BC). Dr Isserow is a clinical assistant professor of cardiology in the Faculty of Medicine at UBC. He is also a staff cardiologist at UBC Hospital and Vancouver General Hospital, and director of the Vancouver General Hospital Centre for Cardiac Rehabilitation.